Navigating the Medicare Transaction Facilitator: A Game-Changer for PBMs, Medicare Part D Plans, and Pharmacies
Posted on October 9, 2025
Operational Excellence is Essential for Plans and PBMs
Key Points
- The Medicare Transaction Facilitator (MTF), fully operational in 2026, requires plans and PBMs to integrate data and payment workflows to support CMS’s negotiated Maximum Fair Prices (MFPs) for high-cost drugs.
- Accurate and timely PDE submissions are critical, as manufacturer refunds, and pharmacy cashflow stability, depend on CMS acceptance.
- Plans and PBMs must update contracts to mandate MTF enrollment, provide reliable SDRA estimates, and implement real-time eligibility checks to minimize PDE errors.
- Operational excellence, including automated PDE pipelines and proactive pharmacy support, will be essential to avoid disruptions and maintain network viability.
In the ever-evolving healthcare landscape, the Inflation Reduction Act (IRA) has brought significant reforms to Medicare Part D, aiming to make prescription drugs more affordable for millions of beneficiaries. A key component of these changes is the Medicare Transaction Facilitator (MTF), a platform managed by the Centers for Medicare & Medicaid Services (CMS) that allows for the implementation of negotiated maximum fair prices (MFPs) for high-cost medications.
For pharmacy benefit managers (PBMs), Medicare Part D plans, and especially independent pharmacies, the MTF presents an opportunity for more transparent pricing but also introduces operational challenges. As we approach complete implementation in 2026, understanding the MTF’s processes, timeline, challenges, and cash flow implications is crucial.
This blog will delve into these aspects, highlighting the operational excellence needed from plans and PBMs to address Prescription Drug Event (PDE) errors and ensure smooth reimbursements, ultimately safeguarding pharmacy viability amid evolving regulations.
A Brief Summary of the MTF Process
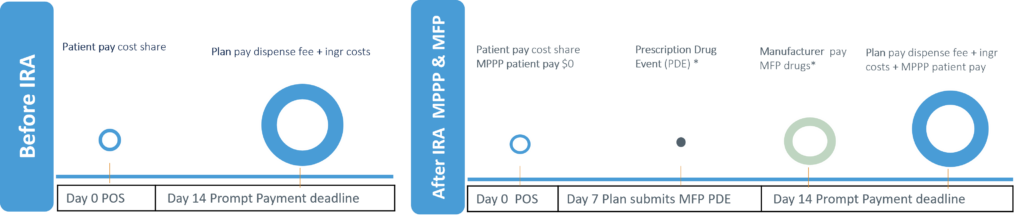
The MTF serves as a hub for data sharing and payment facilitation under the Medicare Drug Price Negotiation Program. It consists of two modules: the MTF Data Module (MTF-DM), which facilitates the exchange of claims information, and the MTF Payment Module (MTF-PM), which handles refund processing. This system shifts away from traditional discount structures, ensuring MFPs are realized without burdening beneficiaries at the point of sale.
The process unfolds as follows:
Pharmacies: Upon dispensing a negotiated drug, pharmacies submit claims to Part D plans at the MFP level, which may fall below acquisition costs. Initial reimbursements come from plans, but pharmacies depend on manufacturer refunds to cover the difference. This difference is typically the wholesale acquisition cost (WAC) minus MFP, known as the Standard Default Refund Amount (SDRA). Enrollment in the MTF-DM is mandatory for pharmacies to access data and receive refunds via electronic remittance or checks.
Manufacturers: September 1, 2025, was the deadline for manufacturers to submit MFP Effectuation Plans to CMS, detailing refund methods. Refunds are issued within 14 days of claim finalization, with transparent reporting to avoid disputes.
PBMs and Part D Plans: These entities update network contracts to enforce MTF enrollment, transmit accurate Prescription Drug Event (PDE) records to CMS, and provide SDRA estimates to pharmacies for better financial planning.
This backend-focused approach prioritizes beneficiary savings (projected at $1.5 billion in 2026) while requiring tight coordination to prevent disruptions.
Timeline of Key Events Through 2026
The MTF’s deployment follows a structured timeline, building on IRA milestones:
| Date/Period | Key Event |
|---|---|
| August 2023 | CMS selects initial 10 drugs for negotiation |
| 2024 | Negotiations conclude; MFPs announced in August 2024 |
| January-June 2025 | Final CMS guidance issued; MTF enrollment opens June 5, 2025 |
| Summer 2025 | MTF 835 Companion Guide published for remittance standards |
| September 1, 2025 | Manufacturers submit Effectuation Plans |
| Fall 2025 | Redacted plans available via MTF-DM; negotiations start for 15 more drugs (effective 2027) |
| November 15, 2025 | Recommended pharmacy enrollment deadline |
| January 1, 2026 | MFPs effective for first 10 drugs; MTF fully operational |
This schedule demands early preparation to avoid last-minute hurdles.
What are the Anticipated Challenges in MTF Implementation?
Implementing the MTF involves technical, administrative, and compliance challenges. Data integration across systems is a primary concern, especially for smaller pharmacies with limited IT resources. Manufacturers may encounter refund calculation disputes, potentially delaying the 14-day window. PDE rejects stemming from errors in beneficiary details or timing could further stall processes, blocking MTF data flows.
PBMs and plans must revise contracts to mandate MTF participation, adding layers of oversight. Variability in drug volumes amplifies risks for high-dispensing pharmacies. CMS continues to refine guidance, but stakeholders anticipate issues like increased administrative loads and beneficiary confusion.
MTF Concerns Related to Pharmacy Cashflow: The Need for Operational Excellence
Pharmacy cash flow emerges as a critical vulnerability under the MFP model. Pharmacies dispense drugs at MFP rates, often below cost, and await manufacturer refunds post-PDE acceptance. Delays in this chain create liquidity gaps and threaten operations, particularly for independent pharmacies which comprise a significant portion of the network.
According to a January 2025 NCPA member survey, pharmacies may face refund delays exceeding 30 days, forcing the average independent pharmacy to float over $27,000 monthly while awaiting reimbursements. With approximately 20,000 independent pharmacies nationwide, this translates to a collective cashflow strain of more than $500 million per month across the sector. High-volume dispensers are hit hardest, as the program eliminates prior margins on these drugs, turning profitable fills into financial drains.
The survey also revealed that 93.2% of independent pharmacists are considering or have decided not to stock one or more of the initial 10 drugs due to these issues. This hesitation could limit patient access, especially in rural areas. Broader financial pressures compound the problem. 96.5% report PBM/plan reimbursements threatening viability, with 80.3% noting declining financial health in 2024.
To mitigate, plans and PBMs must exhibit operational excellence by providing accurate SDRA forecasts, integrating MTF tools for monitoring, and advocating for tailored manufacturer plans. Inventory adjustments and formulary optimizations are essential, but without swift refunds, pharmacies risk closures.
Enhancing Operational Excellence: Focus on Part D Enrollment and PDE Processing
Operational excellence is key to addressing cashflow woes, starting with the Part D enrollment process, where errors often seed PDE issues. Mismatches in eligibility or contract data lead to rejects and can delay refunds.
Plans and PBMs should implement real-time verification via CMS databases, standardize intake with mandatory PDE-linked fields, and conduct audits with pharmacy feedback. Network portals for eligibility checks can preempt problems.
CMS data highlights the stakes. Part D’s FY 2024 improper payment rate was 3.70% ($3.58 billion), often tied to enrollment-related PDE inaccuracies like eligibility edits (e.g., Code 705: beneficiary not enrolled on DOS). FY 2023 saw a similar 3.72% rate ($3.4 billion), underscoring validation needs.
For PDE processing, the 2026 Final Rule mandates 7-day submissions for negotiated drugs to align with 14-day refunds. Excellence requires automated pipelines using the Drug Data Processing System (DDPS), with alerts for rejects. Corrections must occur within 90 days, prioritizing MFP drugs to avoid MTF gaps.
To prevent non-payment complaints, offer upfront SDRA estimates and educate on readjudications. IT investments and training are crucial for handling volumes without errors.
The Unique Dependency on CMS PDE Acceptance
This model introduces a unique shift to the industry. Pharmacies now depend on CMS PDE acceptance for full payment, unlike traditional systems where beneficiary copays were immediate and plan/PBM reimbursements arrived within 14 days, irrespective of PDE status.
With MFPs, initial payments capped at MFP plus fees, and manufacturer refunds hinge on accepted PDEs. Rejects delay data exchange by at least seven days which intensifies cash strains. This backend tie-in is unprecedented and is elevating risks for independent pharmacies, potentially leading to reduced stocking or closures if unaddressed.
Plans/PBMs must prioritize accuracy to protect the ecosystem and foster collaboration for sustainable outcomes.
Looking Ahead: Embracing Operational Excellence for Sustainability
The Medicare Transaction Facilitator is reshaping Part D. The path forward demands strategic adaptation to preserve pharmacy viability and ensure beneficiary access. At PSG, our experts are equipped to help health plans, PBMs, and pharmacies with tailored guidance that drives compliance and operational excellence. From bridging gaps between enrollment and pharmacy teams to streamline PDE reconciliation, to advising on PBM oversight and negotiation strategies aligned with CMS mandates, we help you minimize delays and optimize workflows. Let us guide you to mitigate network pharmacy cash flow complaints through refund acceleration strategies. Let’s turn complexity into opportunity. Reach out today to safeguard your operations and lead the way in sustainable success with PSG.


