Insights into the2019 PBM Formulary Changes
Posted on September 26, 2018
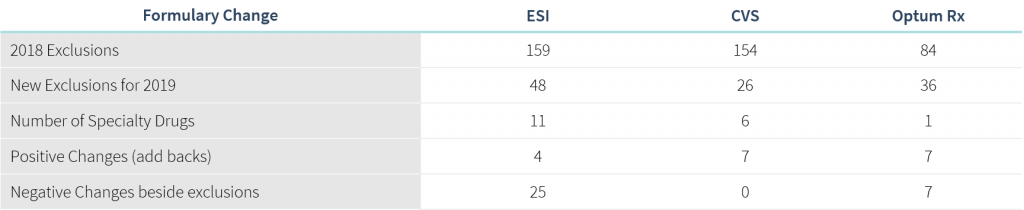
The “Big 3” Pharmacy Benefit Managers (OptumRx, Express Scripts, CVS Caremark) released updates to their 2019 formulary exclusions and this year’s changes come with expansion into some uncharted territories. There is a focus on high-cost drugs with no rebate implications, as well as reversing changes that were made as recently as six months ago in commonly rebated categories. The findings of an impact analysis across our book of business suggest the changes will impact 2.5% of members and 3.3% of total drug spend.
Want to find out how these changes may impact you? Contact PSG today to get the formulary strategy and support you need for an ever-evolving market.
Highlights of the most notable formulary changes are discussed below.
Switching of Preferred Products in Select Categories
- CVS is reversing a 2018 formulary exclusion decision for type 2 diabetes drugs: Jardiance, Synjardy, and Synjardy XR will be added back to the formulary in 2019, while some of the 2018 preferred products within the category — Invokana, Invokamet, and Invokamet XR — will be excluded.
- CVS is changing their only covered test strips — One-Touch — to Accu-Check, where members will need to obtain a new meter and learn any new or different features of the new preferred products.
- OptumRx is adding Rebif, an injectable used for multiple sclerosis (MS), back to the formulary. It was previously excluded July 1, 2018.
- Within the Hepatitis C category, ESI swapped market-share leading, low list price Mavyret for Zepatier, which was previously excluded for 2018. This change may be met with criticism given Mavyret is the most prescribed Hep C drug (47% market share of new prescriptions) and Zepatier the least (1% market share of new prescriptions).
Attention to High-Cost, Low-Value Drugs
- Targeting drugs with no rebate incentives, OptumRx added four key high-cost generic exclusions — Nexium, Zegerid, Fortamet, and Glumetza — in favor of lower cost generic or over-the-counter (OTC) alternatives.
- OptumRx also stands alone among the group as the only PBM to exclude Duexis and Vimovo, two branded drugs which combine two other drugs available OTC. Continued formulary coverage is glaring considering the near-universal clinical opinion that these products deliver few, if any, clinical benefits given that equivalent alternatives cost over $2,000 less per month.
Exclusion of Specialty Drugs in Complex, Rare and Sensitive Therapeutic Categories
- ESI and OptumRx have both chosen to exclude the HIV Atripla. ESI and CVS have both chosen to exclude select hemophilia drugs.
- ESI is the first PBM to consider excluding Berinert, one of four drugs available for the treatment of life-threatening attacks from a rare condition called Hereditary Angioedema (HAE).
- CVS has chosen to exclude Prolastin-C and Zemaira, two products used to treat rare enzyme deficiency conditions.
- Many of these drugs, with the exception of Atripla, are less commonly reimbursed through the pharmacy benefit.
PSG has developed a review of each of the big three PBMs and is providing insights into the changes plan sponsors can expect in 2019, but your PBM may not be represented here. If you would like more information on another PBM, please feel free to contact us at [email protected] to gain further insight into your specific situation.

