Insights into Express Scripts 2022 NPF and Flex Formulary Changes
Posted on August 24, 2021
In recent weeks, Express Scripts (ESI) released their list of January 1, 2022 formulary changes for their National Preferred Formulary and National Preferred Flex Formulary.
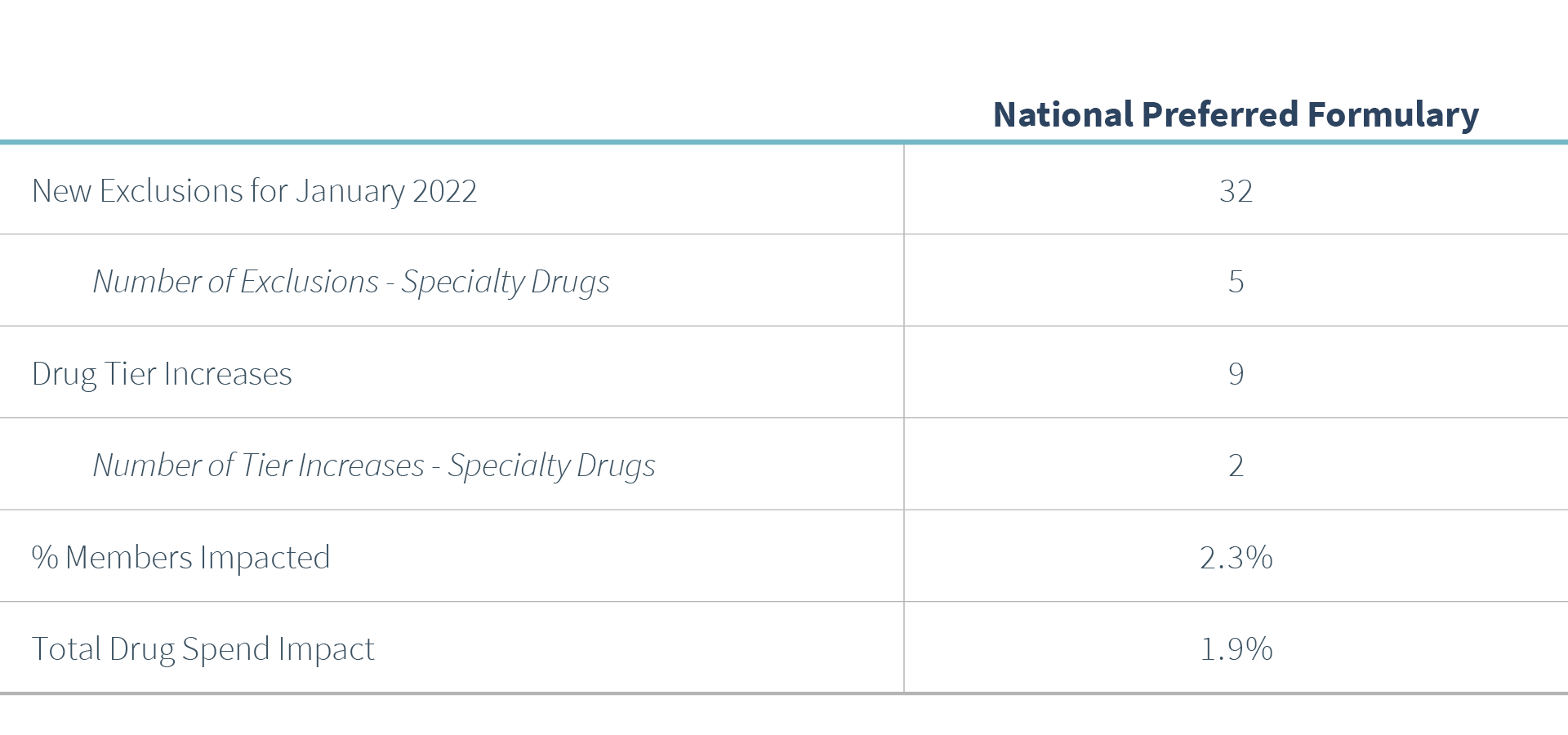
ESI is implementing 32 new exclusions to their National Preferred Formulary, five of which are specialty drugs. ESI is also moving nine medications (including two specialty drugs) to a higher non-preferred tier. Across PSG, clients will experience an average member impact of 2.3% and an average cost impact of 1.9%.
Key Clinical Insights:
- ESI is removing 11 branded medications with a generic equivalent on the formulary, including the highly utilized Synthroid® and Adderall XR®. Together, these two formulary changes impact over half of the members across our book of business (53%).
- The removal of the brand name Synthroid will have the greatest impact at 30.58%.
- Synthroid, used for the treatment of underactive thyroid, is considered a narrow therapeutic index (NTI) drug in which the dose, if not kept even, can result in side effects for patients.
- PSG expects the disruption to be minimal but could be challenging for patients taking brand Synthroid for years.
- In keeping with covering more generics, ESI is creating a generics-only proton pump inhibitors (PPI) category with the exclusion of the last of the branded PPI. (PPIs)
- The drugs included in this exclusionary move include Dexilant, Aciphex Sprinkle, Nexium Packets, and Prilosec Suspension.
- Formulary alternatives will include several generics as well as several PPIs, which are available over the counter (OTC).
- Excluding Dexilant, which is not available as a generic or available OTC, impacts 6% of members across our book of business who will have to transition to a different PPI due to this formulary change.
- However, there are plenty of therapeutically equivalent alternatives available, so member disruption should be minimal.
- ESI is managing the high-cost specialty autoimmune therapies by excluding Remicade and two of its biosimilars: Avsolo® and Renflexis®.
- WIt’s biosimilar, Inflectra®, previously non-preferred, will become the sole preferred agent.
- These drugs are IV infusions, which are often billed through the medical benefit. They are most commonly used to treat rheumatoid arthritis (RA), Crohn’s disease, psoriasis, and other chronic inflammatory conditions.
- In an effort to maintain consistency in drug coverage across benefits for members, plan sponsors may consider covering biosimilars for Remicade under their medical benefit to align coverage where possible.
- Brand Tecfidera® for multiple sclerosis (MS) is another specialty drug exclusion, as a lower-cost generic Tecfidera (dimethyl fumerate) came onto the market in 2020. Tecfidera is the first oral generic treatment for MS available in the US market. This change should result in cost savings for plan sponsors.
- ESI continues to expand its exclusion list of “high cost/low value” drugs, also known as “non-essential drugs.” ESI is doing this with the exclusion of two high-cost brands: Vimovo® and Kerydin®.
- Vimovo® is a combination of the NSAID, naproxen, and PPI, esomeprazole, both available individually over-the-counter.
- Kerydin is used to treat toenail fungus, and similar alternatives are available over-the-counter.
- Many PSG clients have already eliminated these products, which can cost 20 times more than the OTC equivalents.
- While they both have generics now approved, the cost is not much less than the brand. Plan sponsors should consider eliminating coverage of their high-cost generics for more significant plan savings.

