Overview
Over 30% of plan sponsors are exploring alternate models driven mostly by high-cost specialty medications and the need to reduce inappropriate utilization. Right now, 8% have made the leap, but that percentage doesn’t tell the full story. Get the full scoop in this brand-new report.
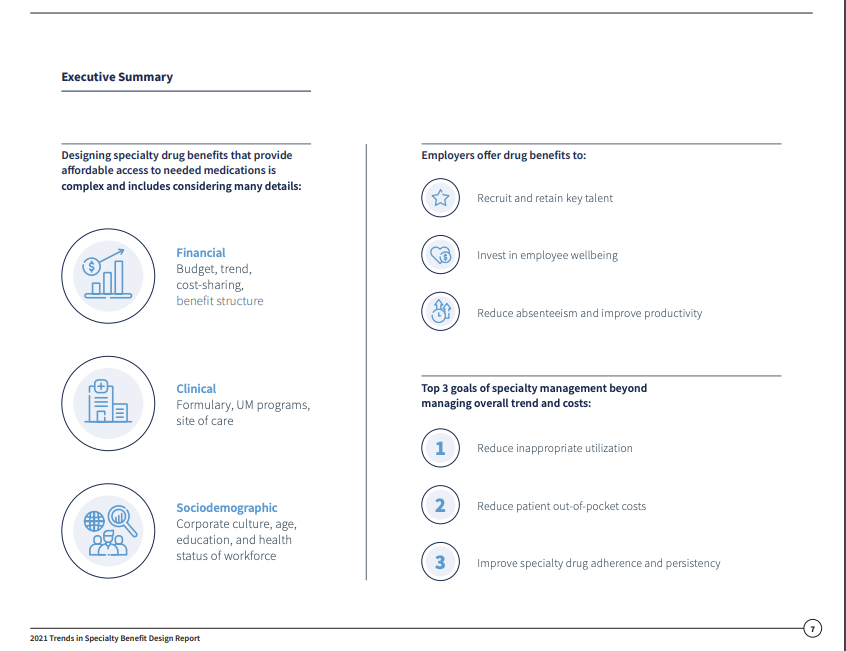
Designing drug benefits that provide affordable access to needed specialty drugs while maintaining an affordable benefit is more complex than ever. Employers offer drug benefits to:
- Recruit and retain key talent
- Reduce absenteeism and improve productivity
- Invest in employee wellbeing
Key Takeaways
- 8% of plans are currently using alternative funding models, while 31% are exploring the use
- 50% of employers are aware of new to market formulary blocks; 20% of plans can track patient treatment delays due to new to market formulary blocks
- Awareness of biomarker testing is low, and only 20% have engaged with their health plan to learn more about coverage options for testing
- 17% of plans feel prepared to make coverage decisions on new disease-modifying Alzheimer’s drugs
- 92% of plans receive rebates under the pharmacy benefit