2023 Formulary Changes: OptumRx
Posted on October 11, 2022
Get the Full Analysis of the Big 3 Formulary Changes:
- Summary of the Big 3 PBM 2023 Formulary Changes
- 2023 Formulary Changes: CVS Health
- 2023 Formulary Changes: Express Scripts
OptumRx has announced formulary changes effective 1/1/2023, organizing them into three themes: affordability, specialty drug updates, and utilization management. This blog is an analysis of these formulary changes and how they may impact plans.
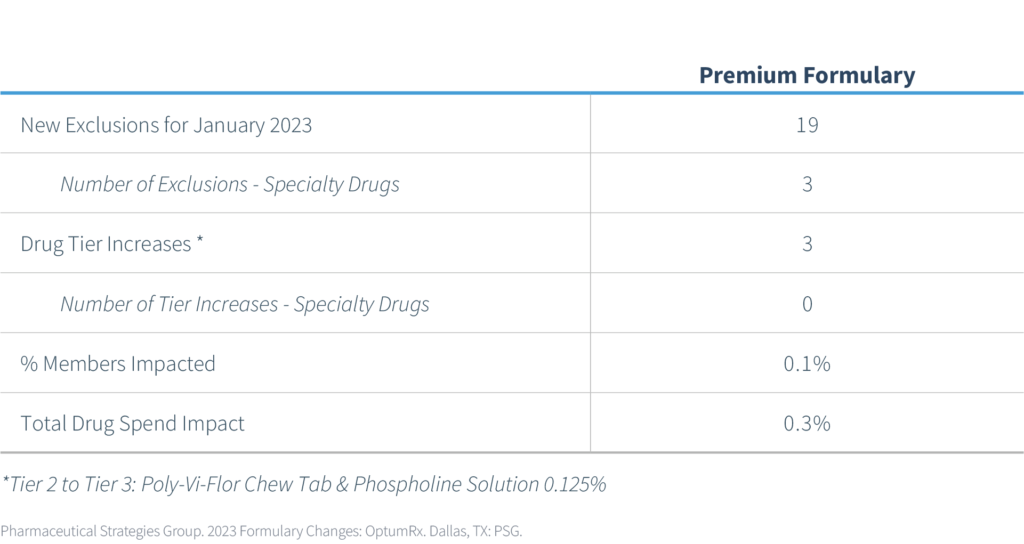
PSG assessed the impact of these changes across our book of business and prepared the following table. Member impact is measured based on the percentage of members from the given PBM currently utilizing one of the newly excluded medications.
Affordability
OptumRx is taking advantage of the FDA approval and availability in the market for 8 new generic drug launches by excluding their brands.
A few examples are as follows:
- Toviaz (fesoterodine fumarate) extended-release tablets for the treatment of overactive bladder
- Combigan (brimonidine tartrate-timolol maleate), a combination eye drop used for the treatment of glaucoma
- Esbriet (pirfenidone) is an oral specialty drug used for the treatment of a rare lung condition, idiopathic pulmonary fibrosis. Important to note, Esbriet is available in both tablets and capsules, but only the Esbriet brand tablets will be excluded.
The exclusion of brand drug Combigan to prefer the generic equivalent represents the largest member impact at 33.5% in our book of business. Additionally, OptumRx is removing several higher-cost brand stimulants to treat attention deficit hyperactivity disorder (ADHD). Some excluded ADHD medicines are unique dosage forms, such as extended-release oral disintegrating tablets (Adzenys XR-ODT, Cotempla XR-ODT), extended-release chewables (Quillachew), extended-release oral suspension (Quillivant XR), and extended-release patch (Daytrana). These higher-cost brand stimulants do not seem to offer additional clinical efficacy over lower-cost alternatives. Consequently, OptumRx has opted to prefer lower-cost generic stimulants (amphetamine-dextroamphetamine IR/ER, dexmethylphenidate IR/ER, dextroamphetamine IR/ER, methylphenidate IR/ER) and a couple of preferred brands (Adderall XR, Azstarys, Vyvanse).
Specialty Drug Updates
For most payers, inflammatory condition drugs are a top therapeutic category of spend for specialty drugs. As many eagerly await the availability of Humira biosimilars in 2023, OptumRx is interestingly moving Enbrel, a long-time brand inflammatory condition specialty drug and competitor to Humira, back to a preferred position. Like Humira, Enbrel is a brand tumor necrosis factor (TNF) inhibitor that shares many of the same indications as Humira. Enbrel also has a few FDA-approved biosimilars, which are not expected to enter the market until 2029.
OptumRx stated, “Enbrel will have preferred status, expanding options within the TNF class and adding value for plans and members.” There are seven FDA-approved Humira biosimilars, with an additional 5 medicines pending FDA approval. Therefore, as many as 12 biosimilars for Humira could launch throughout 2023. With the potential for cost savings in the inflammatory category due to the increased competition with biosimilars, adding a branded alternative such as Enbrel could take market share away from Humira, impacting some of the financial savings plans were expecting.
Utilization Management (UM)
OptumRx stated its goal of improved safety, reduced waste, increased savings, and increased appropriate use through the proposed UM changes. This strategy entails adding new and more restrictive prior authorization, step therapies, and quantity limits to the criteria for these drugs. A notable addition is a new prior authorization for the type 2 diabetes drugs, GLP-1 agonists (e.g., Ozempic, Trulicity), to replace existing step therapy programs. This change is intended to help address the increased utilization of these medications by reducing off-label use for weight loss. OptumRx is reporting a 36% increase in utilization from the previous year, contributing to a 29% increase in per member per month (PMPM) spend in this therapeutic category.
Lastly, OptumRx has decided to move some specialty drugs to non-specialty status, effective 1/1/2023. Pharmacy benefit managers (PBMs) like OptumRx establish proprietary specialty drug lists since there is no industry-wide specialty definition. The non-specialty designation of these drugs will benefit members by allowing additional access through retail and mail pharmacies and the availability of 90-day supplies. In addition, clients may benefit from improved drug discounts for these products as they move away from specialty discounts to retail or home delivery discounts. The UM programs will remain the same despite the status change.
The drugs changing from specialty to non-specialty status include:
- Transplant medicines (e.g., Cellcept)
- Hepatitis B treatments (e.g., Epivir HBV)
- Injectable neurological agents (e.g., Botox)
- Injectable osteoarthritis agents (e.g., Euflexxa)
- Injectable anticoagulants (e.g., Lovenox)

